As we all know, the reason behind using reinforcement in concrete.
Recap – The concrete is strong against compressive force & weak against tensile force. So we use reinforcement bars to increase its tensile strength.
The reinforced concrete is used in construction because of excellent durability, workability & high strength. It doesn’t mean that it will sustain any chemical reactions. The concrete also has some defects and the corrosion is one among them.
What is corrosion of reinforcement in concrete?
The chemical & electrothermal reaction induced by environmental circumstances over a long period which causes the reinforcement bars to get rusted and disintegrated is known as corrosion in concrete.
The reinforcement corrosion severely affects concrete strength & durability.

Corrosion Process in Concrete
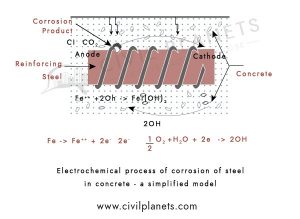
The chemical reaction happens in the following manner,
When two or more materials get in contact, which has distinct surface characteristics such as electric conduit and reinforcement, it forms concentrated cells.
Those concentrated cells will be formed near reinforcement bars due to the differences in the concentrated, dissolved ions like salt, alkalis, acid & chloride, etc. It creates an oxide layer over the reinforcement.
Process
Like an electric battery plus & minus point when two or more materials are used in concrete, it acts as an 
The one end becomes the anode, and the other end becomes the cathode. Then the electrothermal reaction happens in the electrolytic cell.
<— You can skip the next two headings if you are not interested in the chemical formation 🙂 —>
Anode Reaction – The iron cells lose the electrons and pass on the concrete & turn as Ferrous ions.
2Fe → 2Fe2+ + 4e-
Cathode Reaction – The remaining electrons are mixed with water & oxygen, which flow into the concrete.
2H2O + O2 + 4e- → 4OH-
Finally, an oxide layer is formed on the reinforcement & it becomes rust for over a long period. At last, the corrosion affects concrete strength.
What causes concrete corrosion?
If you read the process above, you probably found that without oxygen and water, the corrosion layer does not acquire on the reinforcement.
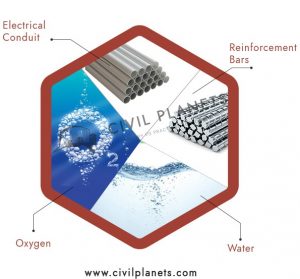
Any defects that allow water or oxygen to enter the reinforced concrete leads to corrosion.
- Water cement ratio – The high water-cement ratio for concrete increases the rate of porosity. So the water molecules enter into the concrete & rust the steel bars.
- Shrinkage in concrete – The shrinkage cracks start to appear on the surface due to the moisture evaporation without any external load. In the future, it exposes the reinforcement bar and gets corroded.
- Quality of Material – The silt or other marine impurities may mix in concrete and develop the cracks on the concrete surface. In the future, the embedded rods are corroded due to its alkalinity.
- Permeability of concrete – The concrete should be poured properly, and compaction is needed to avoid the permeability.
- Moisture – Sometimes, the concrete element may be wet by the surrounding nature. Thus the moisture content may end up to the steel surface through the voids & affect the reinforcement bars.
- Strength of concrete – The concrete structure should be designed well to sustain the load. In the case of low tensile strength or compressive strength of concrete, the cracks can quickly appear on the concrete surface. It makes way to oxygen and water.
- Alkalinity loss due to carbonation – If the reinforcement bars are visible, then the environmental gases react with alkali & make an ion oxide layer on the steel. Finally, the steel will be corroded.
- Sulphate – The cement & other aggregates contain some limited volume of sulphate content. If it exceeds the limitation it may lead to cracks on concrete. To avoid this, the sulphate content in concrete should not be exceeded by 4% by the mass of cement in concrete.
- Chloride – The concrete surface may be contaminated by external environment chloride, such as the salt from the construction material. So the volume of chloride content must be controlled. The quantity of chloride content in the concrete at the time of pouring is given below the table as per the Indian Standard Code IS 456.
| SI | Types of Concrete | Maximum Total Acid Soluble Chloride Content in concrete Kg/Cum |
| 1 | Prestressed Concrete | 0.4 |
| 2 | RCC or PCC containing embedded metal | 0.6 |
| 3 | Concrete not containing embedded metal or any material requiring protection from chloride | 3 |
Effects of corrosion in concrete
- White Patches – The reaction between carbon dioxide & calcium hydroxide makes calcium carbonate. The calcium carbonate penetrates the concrete surface with the help of atmospheric moisture and forms a white patch on the concrete’s external surface. It appears on the bricks also. We have discussed in detail the quality checking of bricks at the site.
- Brown Patches – The brown patches form on concrete surfaces because of the iron oxide layer formed and start corroding the reinforcement bar. After the formation of brown spots, the concrete surface will peel off quickly in some time.
- Cracks – Due to the corrosion, the size of the steel bar may increase (bulge), and on the external surface initially, a minor crack appears. However, after some days, it will become a huge crack which directly affects the stability of the structure.
- Snapping of reinforcement bars – The size of the steel will be reduced due to the continual corrosion. Finally, the steel bars will be snapped, which will affect the concrete element’s alignment.
How do you prevent rebar corrosion on concrete?
- Quality – There is no compromise in the quality of the concrete material. The concrete ingredients should be free from silt or other harmful impurities.
- Clear cover – Sufficient clear cover should be provided for the reinforcement to avoid the chances of exposure.
- Epoxy Coating – The epoxy-coated steel reinforcement shall be embedded in concrete to resist the corrosion.
- Polymeric Fibre – The usage of polymeric fibres in concrete can prevent steel from corrosion.
- Proper Workmanship – Ensure that the concrete should be poured with proper compaction without bleeding and segregation (Honeycomb)to avoid the voids happening on the surface.
Hope you enjoyed the topic. Happy Learning 🙂